ASIA ELECTRONICS INDUSTRYYOUR WINDOW TO SMART MANUFACTURING
Toray Polymer Membrane Extends EV Battery Life
Toray Industries, Inc. has developed an ion-conductive polymer membrane that delivers 10-fold the ion conductivity of its predecessors. This new offering could accelerate the deployment of solid-state batteries1, air batteries2, and other lithium metal batteries. Thus, this greatly expands the cruising ranges of electric vehicles, industrial drones, urban air mobility systems, and other transportation modes.
The transition to electric mobility is increasing demand for lithium-ion batteries delivering higher energy densities. Accordingly, efforts are underway to develop lithium metal batteries whose anodes enable the highest theoretical energy capacity.
Challenges of Lithium Metal
The challenge of lithium metal is its high surface reactivity and the stability issues associated with its dissolution and precipitation morphology during charging and discharging cycles. One notable drawback is the growth of lithium dendrites3, which can cause short circuits. Additionally, metallic lithium anodes in batteries employing solid electrolytes pose similar hurdles, and have yet to see practical applications.
Protective Film for Lithium Metal
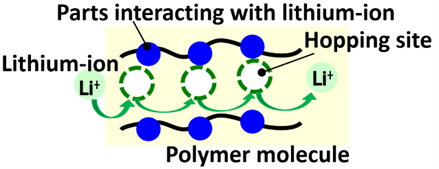
This time, Toray developed polymer membranes offering ion conductivity through hopping conduction. Particularly, this mechanism enables lithium ions to traverse between interacting sites within polymer membranes, effectively jumping across sites. The membranes remain non-porous. This breakthrough leveraged the company’s expertise in molecular design technology, particularly with aramid polymers4, which it refined over many years.
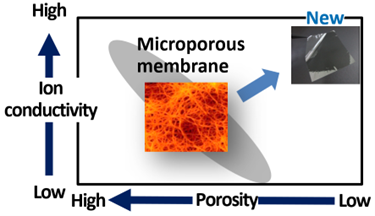
Toray estimates that enhancing the hopping site5 structure and designing a new polymer with more hopping sites has delivered the highest ionic conductivity in the 10-4S/cm range for a hopping-conductive polymer film.
Mainly, the company confirmed that the polymer film functions effectively as a protective film on lithium metal surfaces to overcome the issues mentioned earlier. Moreover, it should extend the service lives of batteries using lithium metal lithium anodes.
Joint research with Professor Nobuyuki Imanishi of the Graduate School of Engineering at Mie University verified the achievement of 100 charge-discharge cycles for the first time in a dual-component lithium-air battery employing this polymer membrane as a separator.
Further, Toray will accelerate research to swiftly establish technology for deployment on solid-state, air, and other advanced batteries.
Part of the development work for the new membrane was through a project funded by the New Energy and Industrial Technology Development Organization (NEDO).
Toray will keep leveraging its core technologies of synthetic organic and polymer chemistry, biotechnology, and nanotechnology to innovate materials in keeping with its commitment to delivering new value and contributing to social progress.
Glossary
1. A solid-state battery uses solid electrolytes instead of the liquid or polymer that lithium-ion batteries employ. The inflammable electrolytes of solid-state batteries enhance safety. Another benefit is that charging is faster.
2. A lithium-air battery is light and offers high capacity. It employs a lithium metal anode and an oxygen cathode. A organic electrolyte anode and aqueous electrolyte cathode structure is under consideration.
3. Lithium dendrites are branch-like lithium crystals that grow when charging batteries. Dendrite growth can degrade battery performance and cause short circuits.
4. An aramid (aromatic polyamide) is a high-performance polymer offering superb heat resistance and rigidity. Toray is the world’s only company to commercialize aramids, through its mictron® film brand. A common application is data storage tapes, which takes advantage of its outstanding rigidity for mass-produced films. Another use is as a circuit material for thin films because its heat resistance ranks second only to that of polyimide.
5. A hopping site refers to specific atoms or atomic groups in polymer chains serving as a transit point for lithium ions to undergo hopping conduction in a polymer membrane.
-22 March 2024-